Aluminum Induction Brazing Advantages
May 17th, 2021

Before we discuss the advantages of induction brazing of aluminum components, a bit of history on aluminum vs. copper within the HVAC/R industry.
Aluminum has been the dominant choice for heat exchangers within the automotive industry for decades. Aluminum heat exchangers, such as radiators, heater cores, evaporators and condensers, used for air conditioning and for engine and powertrain cooling are commonplace replacing copper/brass decades ago.
This material has made similar inroads within the HVAC/R industry. Why has aluminum increasingly become an option to copper? Aluminum is less expensive than copper, it is lighter than copper, and is recyclable. An all-aluminum heat exchanger coil with aluminum tubes and aluminum fins is lighter than copper tubes with aluminum fins. Although aluminum has a thermal conductivity of roughly 60-percent of copper, a pound of aluminum yields nearly three times the parts as a pound of copper giving aluminum a better surface-to-weight ratio.
| Material | Thermal Conductivity [BTU/(hr·ft⋅°F)] |
| Aluminum | 136 |
| Copper | 231 |
| Source: Engineer’s Edge |
Aluminum vs. copper corrosion resistance
Copper coils with aluminum fins are impacted by two types of corrosion, galvanic and formicary. Galvanic corrosion is defined as “an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte” (Wikipedia). Typically, galvanic corrosion doesn’t occur indoors or in dry atmospheres. However, when aluminum in contact with a more noble metal, like copper, particularly in regions with high chloride levels, such as areas near the ocean, corrosion will occur. See Figure 1.
| Material | Density – p`(lb/ft3) |
| Aluminum | 169 |
| Copper | 558 |
| Source: The Engineering Toolbox: The Engineering Toolbox |
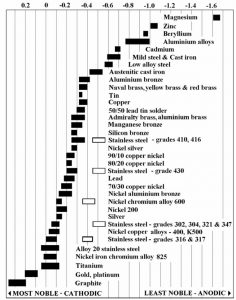
Aluminum HVAC/R coils have another advantage over copper coils – aluminum tubing invulnerability to formicary corrosion. Formicary corrosion occurs out of a chemical reaction between oxygen, water, and organic acid in the coil. Air Conditioning Contractors of America (ACCA) has published an education article, “What Causes Formicary Corrosion & How Is It Stopped?” that discusses in more detail what is formicary corrosion and the common causes. It occurs only within copper-based coils and has been a long-standing problem with copper tubing within HVAC/R application. However, aluminum is immune to formicary corrosion.
Why induction braze vs. torch braze aluminum components?
First, a little background on common aluminum alloys prevalent today and the common aluminum braze and solders used for joining. Brazing aluminum components is much more challenging than brazing copper components. Copper melts at 1980°F (1083°C) and it changes color as it is heated. Aluminum alloys often used in HVAC systems start to melt at approximately 1190°F (643°C) and do not provide any visual cues, such as color changes, as it heats.
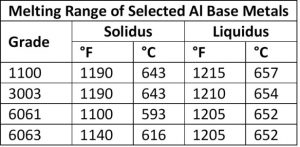
Very precise temperature control is required as the difference in the melting and brazing temperatures for aluminum, dependent upon the aluminum base metal, braze filler metal, and mass of the components to be brazed. For example, The temperature difference between solidus temperature of two common aluminum alloys, 3003 series aluminum, and 6061 series aluminum, and the liquid’s temperature of frequently used BAlSi-4 braze alloy is 20°F – a very narrow temperature process window, thus necessitating precise control. The selection of base alloys is extremely important with aluminum systems that are being brazed. The best practice is to braze at a temperature that is below the solidus temperature of the alloys the make up the components being brazed together.
| AWS A5.8 Classification | Nominal Chemical Composition | Solidus °F (°C) | Liquidus °F(°C) | Brazing Temperature |
| BAISi-3 | 86% Al 10%Si 4%Cu | 970 (521) | 1085 (855) | 1085~1120 °F |
| BAISI-4 | 88% aL 12%Si | 1070 (577) | 1080 (582) | 1080~1120 °F |
| 78 Zn 22%Al | 826 (441) | 905(471) | 905~950 °F | |
| 98% Zn 2%Al | 715(379) | 725(385) | 725~765 °F |
It should be noted that galvanic corrosion can occur between zinc-rich areas and aluminum. As noted in the galvanic chart in Figure 1, zinc is less noble and tends to be anodic compared to aluminum. The lower the potential difference, the lower the rate of corrosion. The potential difference between zinc and aluminum is minimal compared to the potential between aluminum and copper.
What is Pitting?
Another phenomenon when aluminum is brazed with a zinc alloy is pitting. Local cell or pitting corrosion can occur on any metal. Aluminum is normally protected by a hard, thin film that forms at the surface when they are exposed to oxygen (aluminum oxide) but when a flux removes this protective oxide layer, dissolution of the aluminum can occur. The longer the filler metal remains molten, the more severe the dissolution is.

Aluminum forms a tough oxide layer during brazing, so the use of flux is essential. Fluxing aluminum components can be done separately prior to brazing or an aluminum brazing alloy containing flux can be incorporated into the brazing process. Depending upon the type of flux used (corrosive vs. non-corrosive), an additional step may be required if the flux residue must be removed after brazing. Consult with a braze and flux manufacturer to get recommendations on brazing alloy and flux based on the materials being joined and the expected brazing temperatures.
The advantages of induction brazing of aluminum vs. torch or flame brazing
The low melting temperature of aluminum base metals coupled with the narrow temperature process window of the braze alloys used is a challenge when torch brazing. The lack of color change while heating aluminum does not provide braze operators any visual indication that the aluminum has reached the proper brazing temperature. Braze operators introduce a number of variables when torch brazing. Among these include torch settings and flame type; distance from torch to parts being brazed; location of flame relative to parts being joined; and more.
Reasons to consider using induction heat when brazing aluminum include:
- Quick, rapid heating
- Controlled, precise heat control
- Selective (localized) heat
- Production line adaptability and integration
- Improved fixture life and simplicity
- Repeatable, reliable brazed joints
- Improved safety
Successful induction brazing of aluminum components is highly dependent on designing induction coils to focus the electromagnetic heat energy into the areas to be brazed and to heat them uniformly so that the braze alloy melts and flows properly. Improperly designed induction coils can result in some areas being overheated and other areas not receiving enough heat energy resulting in an incomplete braze joint.

For a typical brazed aluminum tube joint, an operator installs an aluminum braze ring, often containing flux, on the aluminum tube and inserts this into another expanded tube or a block fitting. The parts are then placed into an induction coil and heated. In a normal process, the braze filler metals melt and flow into the joint interface due to capillary action.
Radyne can help customers optimize their brazing processes. Aluminum being rather complex to braze, Radyne with its IPEC lab can model the braze process and conduct brazing experiments to improve brazing efficiency and optimization of the brazing process.
Conclusion
Not all brazing applications are strong candidates for induction heating. In situations of low volume, or high mix, or irregular shapes where fixturing or the coil design would be extremely complex, furnace brazing or torch brazing with experienced operators may be a more economical choice. However, Induction brazing allows for fast, controllable, precise, energy-efficient, repeatable braze joint for companies that embrace lean manufacturing.
As an induction heating industry leader, Radyne can help you navigate the challenges and realize the advantages of induction brazing. For more information on solutions for induction heating and related processes, please contact us.




